This is Part 2 in a blog series about how I teach Structure and Bonding at KS4. Part 1 discussed the teaching sequence I used to follow and the main challenges and misconceptions presented in this topic.
Before introducing anything new about bonding types and the resulting structures, I spend time reviewing the particle model and states of matter. One of the key concepts students need to understand is why different substances (and hence structure types) have different melting and boiling points – this seems to be the property which they are most likely to write muddled explanations for, so I start with a thorough recap framed in a way which will then be mirrored as I introduce each new structure type during the course of the topic.
I begin with the macroscopic view and will approach each structure from this perspective – zooming in from the familiar and concrete to the abstract. This is the complete opposite of what I used to do teaching the bonding and then zooming out to the macroscopic structure. I have found that starting with something concrete which students are completely familiar with enables them to build the structure-property links much more readily.
My lesson goes something like this:
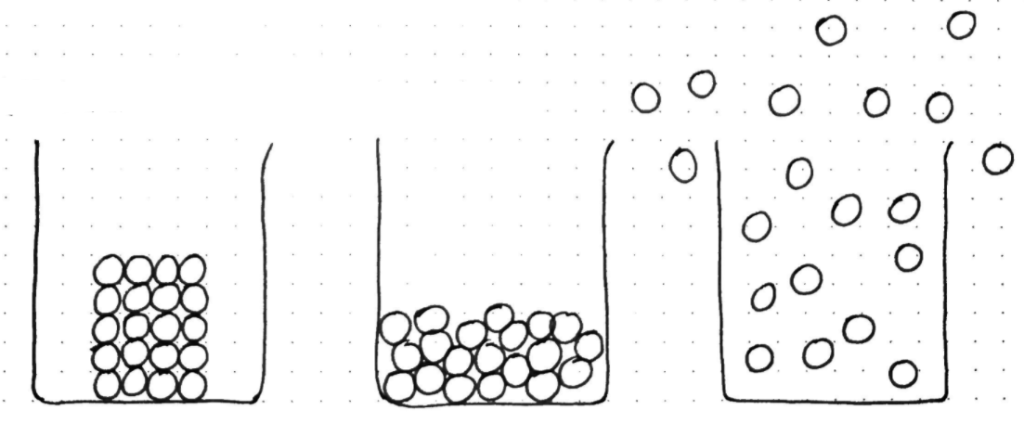
Showing these images under the visualiser, I ask what they represent?
Students will usually immediately recognise solid, liquid and gas (or perhaps ice, water and steam). I have deliberately drawn these in beakers, not in boxes as they are often shown in textbooks, as it immediately makes the example more concrete and relatable to the students’ own real life experience.
I then label the images and ask what is required to turn a solid into a liquid and a liquid into a gas. Students might answer, “heat”. I would then ask what form of energy this is, looking for the answer, “thermal energy”. Finally at this stage, I would check that students can remember the terms, melting, boiling, freezing and condensing. All the while I’m building up the annotations on the diagram:
Although I would check up on students’ knowledge of freezing and condensing, I’ve only added the melting and boiling labels to the diagram as in this topic we will be focusing on explaining melting and boiling points.
At this point I introduce the terms macroscopic and microscopic, explaining that what we’ve been discussing so far is what is happening at the macroscopic level which we can see and experience with our senses. I then explain that chemistry is about understanding processes and interactions at the microscopic level – What is going on inside the solid, liquid and gas? What are they made up of? Why do things melt? Why do different substances melt at different temperatures?
Next I ask students to draw the particle diagrams for a solid, a liquid and a gas on mini whiteboards – this gives me opportunity to see what each student has remembered and whether they have any misconceptions about the arrangement of particles (particularly in liquids) from KS3. We’ll then discuss the good examples and draw out any learning points from those which are not quite right.
At this point I will model drawing particle diagrams under the visualiser and get students to copy these into their books so that they are all practising drawing accurate diagrams, once armed with the knowledge of mistakes to be avoided:
Again, I draw these diagrams in the beakers to make stronger links with the macroscopic representation we’ve just been considering. I also think it’s important to stress that there will be the same number of particles in each diagram (I’ll ask questions to check students’ understanding about this) and to link this to conservation of mass and the fact that the particles themselves are not changing. I’ve purposely drawn the gas particles escaping from the beaker as this is something else which I find students often don’t grasp when particle diagrams are drawn with the particles confined to boxes.
Next I’ll ask questions about how a substance goes from solid to liquid and liquid to gas – probably cold-calling one of the weaker students in the class as this is something we have already covered in the lesson. I’ll insist on them using the correct terminology of thermal energy, then I’ll add these labels to my diagram once more relating it back to the macroscopic version.
Now it’s time to really start thinking about what’s going on at the microscopic level.
I always start with the solid and ask, “How would you describe the arrangement of particles in a solid?” Students will usually respond by saying that they’re arranged in a “regular pattern” or a “rigid structure” as they have learned this at KS3. I’ll then ask, “What’s holding the particles together?” The answer is usually “forces” or “bonds”. I explain that these answers are true, but that I’d like students to describe these as “interactions” because the words forces and bonds have specific meanings and it’s important that we use scientific vocabulary correctly.
At this point I will annotate the solid particle diagram to show that the interactions between the particles are holding them in a rigid structure.
We then move onto the liquid diagram. “How are particles in the liquid arranged?” “What holds the particles in a liquid together?” Here, I want students to recognise that the particles in the liquid are still held together by the same interactions as those in the solid. I then explain that the reason the particles are now free to move around (enabling the liquid to flow and take the shape of its container) is because some of the interactions have been broken or weakened. I’ll ask, “What has broken or weakened these interactions?”, it’s the thermal energy which has transferred to the kinetic energy store of the particles enabling them to partially overcome the interactions which held them tightly together in the solid.
I’ll then add annotations to the liquid diagram which students can copy on their own.
Lastly, we discuss the gas. “How are the particles arranged in a gas?” I explain that the particles are no longer held together, but have all become separated from one another. I ask students to explain why and we link it to the thermal energy which has been transferred to kinetic energy in these particles enabling them to completely overcome the interactions between the particles leaving them to move around freely and rapidly. With a high ability class I might discuss how much energy is needed for boiling compared to melting and link this to the fact that specific latent heat of vaporisation is generally much greater than that of fusion, due to the fact that all the interactions are being broken, rather than just some.
The annotated diagram will now look like this:
Finally, I want students to be able to explain why different substances have different melting and boiling points as this is going to be key for them in understanding the influence of structure and bonding types on these properties.
I want all students to be really thinking about this. Using cold-call a lot in lessons ensures that they do, but for a more challenging concept like this, I’d probably use think, pair, share to enable students to develop their answers before I select someone to share. The questions I ask are, “If I put a beaker containing a block of ice on a tripod above a Bunsen burner, what happens?” The ice melts. “Does the glass beaker melt?” No. “Do the metal tripod and gauze melt?” No. “Thinking about what you’ve learned about interactions between particles and melting, explain why the ice melts but the glass and metal don’t melt.” I then give thinking time, a short time for students to share their answers giving them all practice in articulating their thoughts to others, and then ask a student to share their answer.
We then develop this together as a class until we reach something like, “When heated with a Bunsen burner a block of ice in a beaker melts because the thermal energy transfers to kinetic energy in the particles in the solid, weakening the interactions and allowing the particles to move around and the liquid to flow. The glass beaker and the metal tripod and gauze do not melt because the interactions between their particles are stronger and require a greater amount of thermal energy for them to be overcome. If we were able to heat them to a higher temperature then they would melt.”
The last thing I get students to put down in their notes is that some substances have higher melting and boiling points that others because the interactions between their particles are stronger and require more thermal energy to overcome them.
I’ll then ask students to write down their answer to the question about the ice in a beaker, from memory, before completing some SLOP work to consolidate learning.
I’m now confident that my students are ready to tackle structure and bonding because I have introduced the macroscopic and microscopic representations which they will need to be able to switch between constantly, and they have a secure grasp of the particle model which I can use as the basis for introducing each structure type – “What do the particles represent?” and “What are the interactions between them?” are questions I will keep coming back to when introducing each structure type and discussing its properties.
Although everything I’ve taught in this lesson should be review of prior knowledge, I’ve found that it’s really worth taking the time to crystallise students’ understanding of this, and that doing so helps to make the links between structure, bonding and properties much more tangible.
The teaching sequence I then follow is:
- Ionic structures, bonding and properties
- Metallic structures, bonding and properties
- Giant covalent structures, bonding and properties
- Simple covalent structures, bonding and properties.
I plan to write about how I teach the key areas for each structure, I’ll link them here when they’re ready.
5 thoughts on “Teaching Structure and Bonding (Part 2): Laying Foundations – The Particle Model”